What Mitochondria Actually Do And Why You Feel It When They Decline
A Simple Breakdown of What's Actually Happening Inside Your Cells
You already know the line.
“Mitochondria is the powerhouse of the cell.”
Every person who took biology in middle school has that burned into their brain. Teachers love saying it. Memes keep it alive.
And it tells you almost nothing useful.
Because if the mitochondria are the powerhouse, what does that actually mean for how you feel at 44? What does it mean for why your recovery takes longer than it used to? Why you hit a wall at 2pm every day even when you slept 8 hours?
Nobody connects those dots. So let me do it.
What Mitochondria Actually Are
Your body is made of roughly 37 trillion cells. Most of those cells have mitochondria living inside them. Your heart cells might have 5,000 mitochondria per cell. Your muscle cells, thousands more. Your brain cells are loaded.
They earned the “powerhouse” label because they produce ATP. ATP is the energy currency your body runs on. Every time you lift a weight, think a thought, pump blood, or digest food, ATP is what makes it happen. Without ATP production, nothing works.
But calling mitochondria just an energy factory misses most of what they do.
Mitochondria also control apoptosis, which is programmed cell death. When a cell gets damaged beyond repair, mitochondria are the ones that signal it to shut down. That is a good thing. You want damaged cells eliminated before they cause problems.
They regulate calcium signaling, which affects muscle contraction and nerve transmission. They produce heat to maintain body temperature. They generate reactive oxygen species, which in small amounts are useful signaling molecules but in excess become the oxidative stress that damages tissue and accelerates aging.
And they communicate with your cell’s nucleus, influencing gene expression. Your mitochondria are sending signals that affect which genes get turned on and off.
This is not a simple power plant. It is closer to a command center.
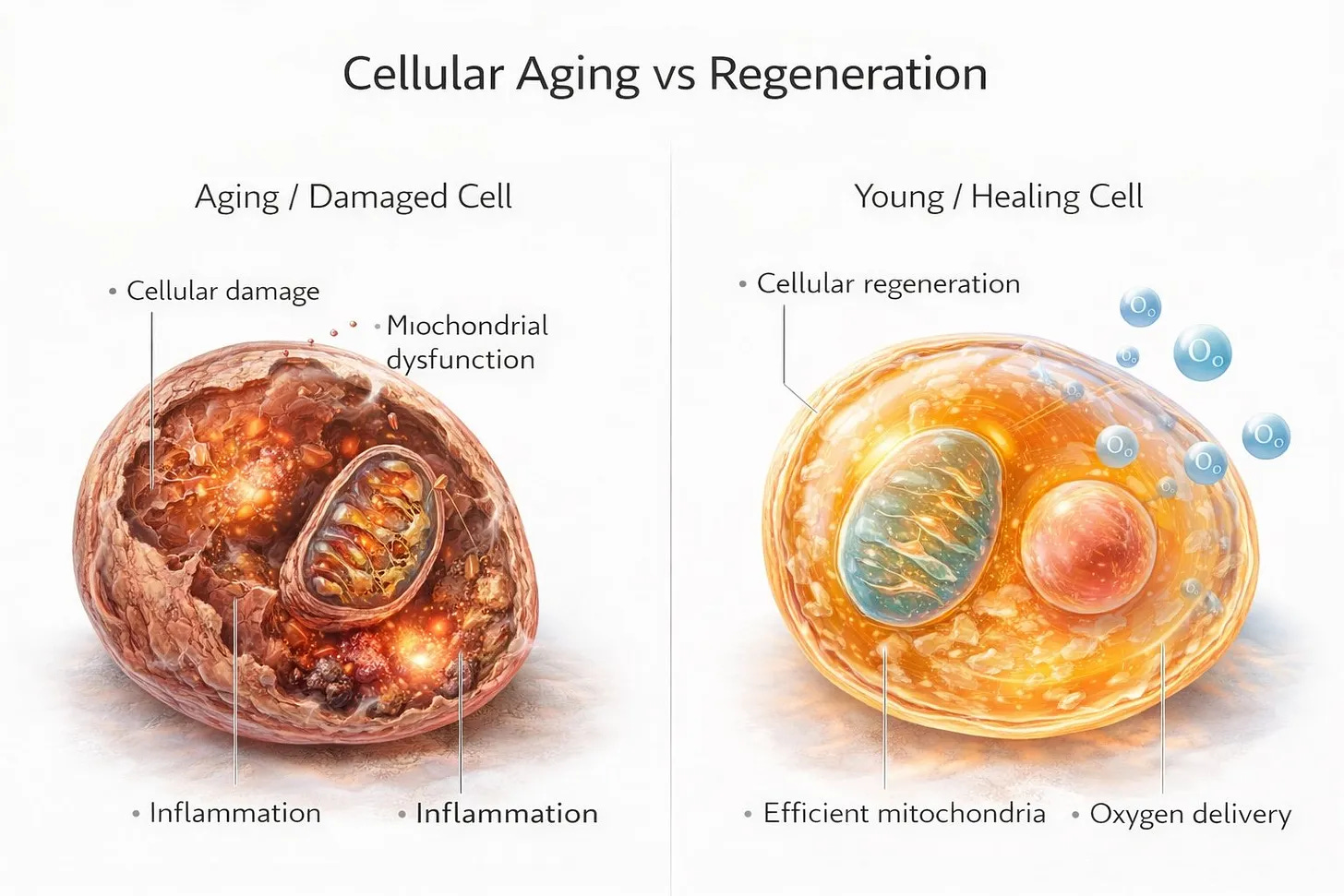
What Happens When You Age
Around your mid-30s, mitochondrial function starts declining. By 40 it is measurable. By 50 it is significant. By 60, some research suggests you have lost roughly half of your peak mitochondrial capacity.
Two things happen simultaneously.
First, you produce fewer mitochondria. The process that creates new mitochondria, called mitochondrial biogenesis, slows down. Your cells stop replacing old, worn-out mitochondria as efficiently. You end up with less total mitochondrial mass.
Second, the mitochondria you have become less efficient. The machinery inside them accumulates damage. They produce less ATP per cycle. They generate more oxidative stress as a byproduct. The signal quality degrades.
Fewer mitochondria. The ones that remain are worn down and underperforming. That combination is what makes 45 feel nothing like 25.
Now map that to real symptoms.
The afternoon energy crash that did not happen in your 20s is mitochondrial output declining. Recovery taking 3 days instead of 1 is mitochondria unable to keep up with cellular repair demands. Brain fog and slower thinking happen because your brain is your highest-energy organ and is the first to feel the drop. Stubborn body fat, especially around the midsection, is mitochondrial dysfunction directly impairing fat oxidation and metabolic efficiency.
Feeling like you are running on 70% no matter how well you sleep or eat is not in your head. The cellular infrastructure is degrading.
This is why aging feels the way it feels. Not one dramatic moment. A slow erosion of cellular energy production happening in trillions of cells simultaneously.
The Fuel Problem: NAD+
Your mitochondria run on a coenzyme called NAD+, nicotinamide adenine dinucleotide.
Think of NAD+ as the fuel your mitochondria need to actually function. Without it, the machinery stalls. Energy production drops. The repair pathways go offline.
NAD+ does three critical things.
It powers the electron transport chain, which is the actual process inside mitochondria that converts food into ATP. No NAD+, no ATP.
It activates sirtuins. Sirtuins are proteins that regulate DNA repair, inflammation control, and cellular longevity. They are sometimes called longevity proteins. They require NAD+ to function. Without NAD+, your sirtuins are essentially sitting idle.
It supports PARP enzymes, which repair DNA damage. Every day your DNA takes hits. PARP repairs it. PARP runs on NAD+.
Your NAD+ levels drop roughly 50% between age 40 and 60. The same decade your mitochondrial function is already declining, you are also losing the primary fuel your mitochondria need to run.
You cannot fix mitochondrial function without addressing NAD+ first. It is the foundation everything else builds on.
One thing worth understanding before looking at dosages is why NAD+ protocols use a loading phase.
If your levels have been depleted for years and you start at a low maintenance dose, you are dripping fuel into a tank that is running on empty. It works eventually but it takes a long time to feel anything. The loading phase is how you fill the tank fast. You dose daily for 7-10 days to saturate your cells with NAD+ as quickly as possible. Once levels are restored you drop to a maintenance schedule because now you are topping off a full tank instead of filling an empty one.
Most people who skip the loading phase start a protocol, feel nothing after two weeks, and quit. The loading phase is what gets you over the threshold fast enough to actually notice a difference.
Research protocol context: In research settings, injectable NAD+ is studied using a two-phase approach. The loading phase uses 100-200mg subcutaneously daily for 7-10 days to saturate depleted cellular reserves. After the loading phase, maintenance drops to 100-200mg SubQ administered 2-3 times per week. Many researchers run maintenance continuously, adjusting frequency based on response. Doses above 200mg are associated with increased side effects including nausea and flushing and are not recommended for SubQ self-administration. Injectable NAD+ bypasses the conversion steps required by oral precursors like NMN and NR, delivering the compound directly.
The Building Problem: MOTS-c
NAD+ restores and fuels your existing mitochondria. That is essential. But it does not solve the other half of the problem, which is the fact that you have fewer mitochondria than you used to.
That is where MOTS-c comes in.
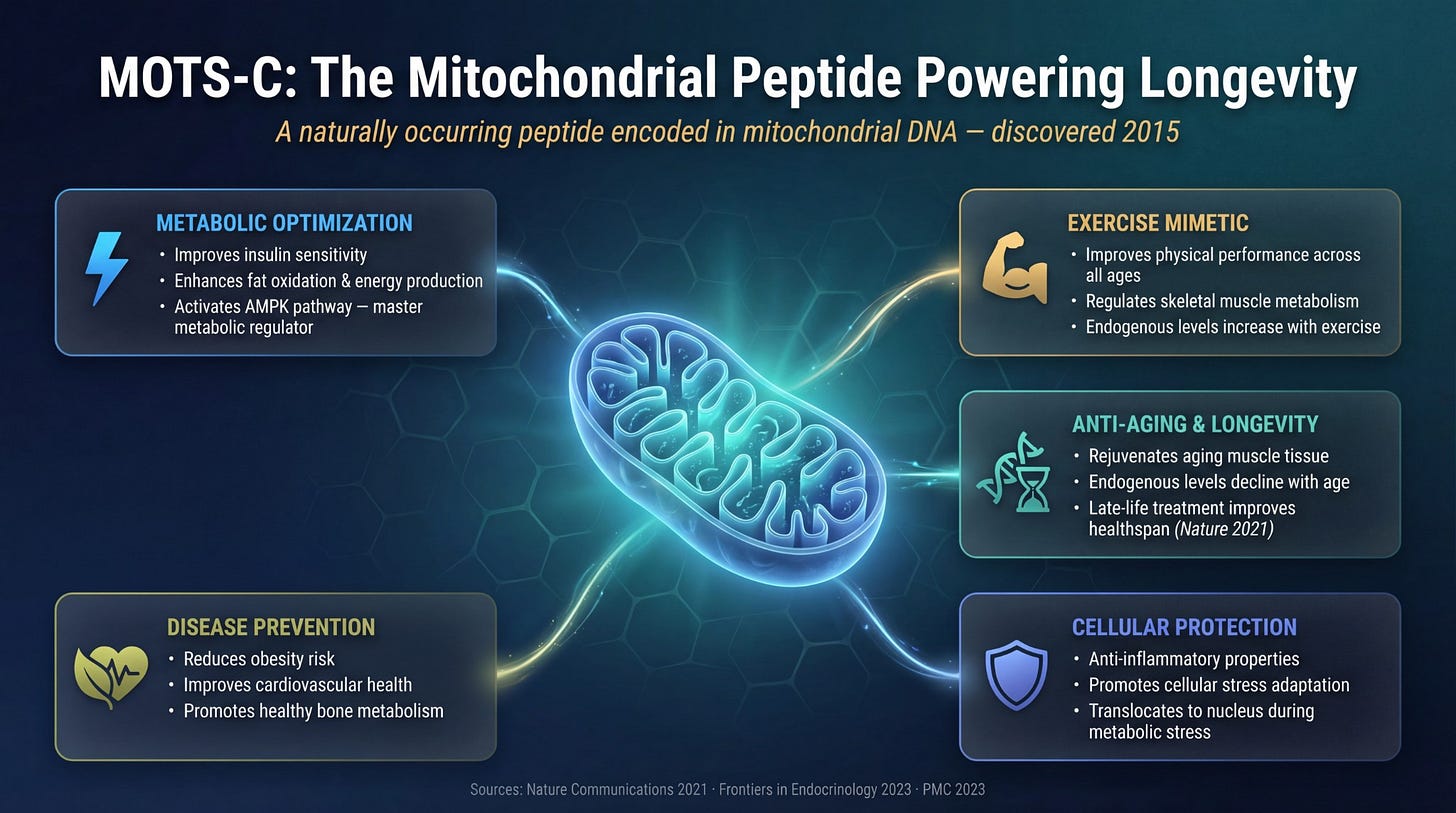
MOTS-c is a mitochondrial-derived peptide, first identified in 2015. It activates something called AMPK, AMP-activated protein kinase. AMPK is a master metabolic regulator. When AMPK gets activated, a cascade of things follows.
Your body signals mitochondrial biogenesis, the creation of new mitochondria. You are not just working with what you have. You are building more.
Insulin sensitivity improves. Your cells take up glucose more efficiently, which feeds directly into better energy production. Fat oxidation increases. Your body gets better at burning fat as fuel. Inflammation decreases at the cellular level.
Some researchers describe MOTS-c as mimicking the cellular response to intense exercise, specifically the mitochondrial growth signal you get from hard training.
Research protocol context: In research settings, MOTS-c is studied at 5mg subcutaneously, 3 times per week, administered in the morning. Most protocols run 10 weeks on followed by a break period of 4 to 6 weeks before resuming.
MOTS-c is a research peptide. Human studies are still limited. What we understand comes primarily from animal research and early human data. This is for research purposes only.
Why These Two Together
NAD+ and MOTS-c address mitochondrial decline from opposite directions.
NAD+ works on what you have. It restores the fuel supply, activates the repair proteins, and brings existing mitochondria back toward peak function.
MOTS-c works on what you are building. It signals your body to create new mitochondria and improves how efficiently those mitochondria operate.
Together they cover both sides of the problem. More mitochondria, all running with the fuel and repair systems they need to actually perform.
This is why the combination has become a focus in longevity and biohacking research. Not because either compound is a cure. Because the two mechanisms are complementary in a way that neither achieves alone.
The Takeaway
Mitochondria are not just energy factories. They regulate cell death, inflammation, metabolism, gene expression, and aging at the cellular level.
They decline with age, fewer in number and less efficient in function, and that decline maps directly to how most people feel in their 40s and beyond.
The fuel problem is real. NAD+ depletion is measurable and significant.
The building problem is real. Mitochondrial biogenesis slows and new mitochondria stop replacing old ones fast enough.
Both problems have research-backed approaches worth understanding.
I will keep breaking this down as the research develops.
If you want to go deeper on mitochondrial protocols, dosing questions, and what I am personally running, that conversation is happening in my Skool community. It is also where I share the vetted suppliers I actually use and trust If you are serious about this stuff, that is where you want to be.
Join here: https://www.skool.com/peptides-biohacking-hub
— Joe Mars
All content on this Substack is for educational and informational purposes only. Nothing here constitutes medical advice. All peptides discussed are research compounds.